Revance NYC
What is Revance Therapeutics?
Revance is a new aesthetic company, based in Silicon Valley, who is at the forefront of new innovations in neuromodulators for the aesthetic market. Revance has created an advanced neuromodulator product, DaxibotulinumtoxinA (DAXI) for cosmetic and medical indications. They have developed a new longer lasting neurotoxin, which will be a strong competitor to Botox. DAXI has been showed in clinical trials to last much longer than Botox, lasting for nearly six months in duration. DAXI received its first FDA approval on February 6, 2020 to treat glabella (frown) lines. Since neuromodulators are most common noninvasive cosmetic procedure performed in the United States, with 2.6 million total facial injectable procedures performed in 2018 alone, this two billion dollar industry is incredibly competitive.
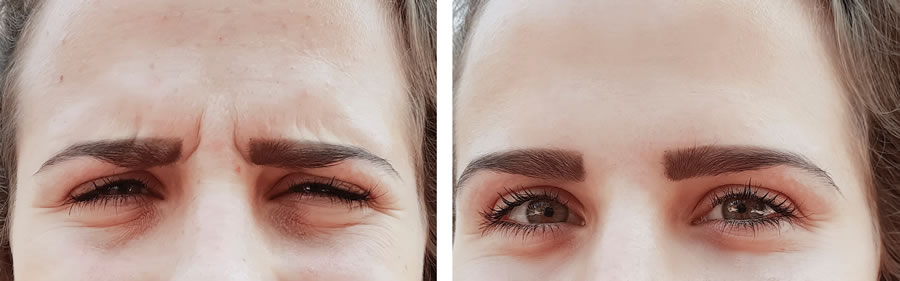
How does DAXI compare to Botox?
The key advantage of DAXI over Botox, which is manufactured by Allergan, is its long-lasting neuromodulator capability. Almost three decades ago, botulinum type A (Botox) was FDA approved for the glabella area. Botox has been extremely popular and it is currently the number one injectable in the United States. However, one of the main complaints with Botox is its short duration of therapeutic effect. The cosmetic effect of Botox typically lasts 3 to 4 months in duration and then dissipates. Patients are extremely interested in a new neuromodulator whose effects would last for a longer period of time. With DAXI lasting twice as long, patients would only need to come twice a year for wrinkle injections.
What other clinical indications will DAXI have?
Revance currently has 12 ongoing clinical trials using DAXI for the treatment of forehead lines, crow’s feet, full upper cervical lines, cervical dystonia, plantar fasciitis and upper limb spasticity.

How long will Revance’s DAXI last?
Current studies of DAXI show 24-week duration in the glabella area. Similar longevity and results can be expected shortly for other areas of the face.
What is TEOXANE SA’s Resilient Hyaluronic Acid (RHA)?
Teoxane SA Resilent Hyaluronic Acid (RHA) is an FDA approved line of dermal fillers, distributed by Revance Therapeutics, for the correction of dynamic facial wrinkles and folds. RHA is a new hyaluronic acid, dermal filler, which will compete with Juvderm and Restylane to treat smile lines (nasolabial area) cheek, chin, and malar area. RHA will be a great new addition to hyaluronic acid fillers.

How do I get started with the new Revance long lasting neurotoxin?
Dr. Michele Green, based in NYC’s Upper East Side, is an internationally re-known dermatologist in the field of cosmetic dermatology. With an expertise in minimally invasive cosmetic injections and lasers, it is best to call to consult Dr. Green at 212-535-3088 or contact us online today.
 212-535-3088
212-535-3088 



